Digital & Computational Pathology Market Report – Third Edition: 2026 - 2031
This report provides an overview of the digital and computational pathology market (referred to within this report as the digital pathology market) from 2026 to 2031. The digital pathology market, as defined in this report, is comprised of revenue from instruments and software sold to clinical, academic, biopharma, and CRO customers, along with algorithm development services and other service revenues generated by CROs and reference labs.*
This market remains highly fragmented, with leading vendors offering increasingly integrated, end-to-end digital pathology platforms spanning slide scanning, visualization, image management, and analytics. In parallel, a long tail of mid- and smaller-sized players continues to focus on point solutions, particularly within AI-driven image analysis. As computational pathology matures and customer needs become more clearly defined, consolidation is expected to accelerate, especially across algorithm and AI tools. Larger platform players are actively expanding capabilities through partnerships, acquisitions, and the development of foundation models, placing growing pressure on standalone AI vendors.
For this report, we leverage a combination of secondary research (e.g., analysis of quarterly and annual reports, earnings call transcripts, clinical trials, congress abstracts, company websites, etc.) and primary research (e.g., 21 interviews with digital pathology stakeholders from hospitals, academia, biopharma, and CROs, 301 clinical pathology lab survey respondents) to assess the current and expected future adoption and utilization of digital pathology tools and technologies, and to characterize the key market dynamics. From our research and analysis, we estimate that the digital pathology market is ~$1.2B in 2026 and is expected to grow at 12% p.a. in the next 5 years, to reach ~$2.1B by 2031.
For each of the subsegments analyzed, we present forecasted data from 2026-2031. In addition, we detail market drivers (e.g., open platforms for 3rd-party integrations) and moderators (e.g., fragmented regulatory policies), market trends (e.g. growing importance of foundation models for discovery and insights), a high-level customer breakdown (clinical use (AMC & community hospitals), large pharma, small pharma, CROs), and offer key information on top competitors. All analyses are based on publicly reported data and DeciBio research and analysis.**
*Reagents and wet-lab materials used for tissue analysis are excluded from the scope of this report, as they are captured in our Spatial Biology Market Report. The scope of the report also excludes digital cytology platforms and technologies.
** Disclaimer: Some of the companies listed in this report may be DeciBio Consulting clients and customers
Technologies and Revenues Included:
- Digital pathology / image analysis in which automated image analysis, algorithms, or pathologist-supported software is used to perform image analysis of any sort from an image of a scanned slide
- 3rd Party Image Analysis performed by a standalone image analysis company or spatial image analysis performed by a standalone image analysis company
- Technology classes which are designed to profile and analyze biomarkers including IF, IHC, H&E, and ISH
- Instrument and software revenues associated with the purchase of digital pathology platforms (e.g., slide scanners) or software used to run the assays and analyze the data, and service revenues associated with the processing or analysis of slides (e.g., by CROs / Reference Labs) or CDx development
- Global sales of products and services to academic, clinical, biotech / pharmaceutical companies, and CRO customers
Technologies and Revenues Excluded:
- Instrumentation, assays, and reagents upstream of slide digitization (e.g., autostainers, dyes, and antibodies), including spatial biology instrument and reagent revenues
- Spend downstream of image analysis (e.g., data storage and compute is assessed qualitatively but not included in the model)
- Spatial analysis conducted on-platform or offered by instrument manufacturers (e.g., analytics tools provided by Bruker, 10x)
- Digital cytology platforms and technologies
- Service revenues passed to CROs / reference labs for slide staining
- Service revenue related to CDx development
Updated Content
- Incorporated the latest trends in the Digital & Computational Pathology market, based on interviews (N = 21) with experts, a clinically-focused survey (N =301) and extensive secondary research
- Updated our database of Digital & Computational Pathology companies and their offerings, highlighting key recent activity and product updates
- Updated market sizing, segmentation, and forecast based on primary research findings and publicly-available information
- Evaluated emerging, high-growth applications in depth, elucidating both quantitative and qualitative market drivers, moderators, and trends, including a new deep-dive on the computational pathology market landscape
Updated Numbers
- Updated a bottom-up market model (based on number of customers by type and annual spend), informed by primary and secondary research (e.g., public filings)
- Assessed market sizes for each of the market segments analyzed, including by customer, products / services, technology, application, therapeutic area, and geography
- Projected revenues for each market segment from the current year (2026) through 2031
Table of Contents
- Introduction & Methodology
- Executive Summary
- Market Analysis
- Computational Pathology Market Landscape
- Partnership Landscape
- Primary Market Reseach (data from 301-respondent clinical survey)
- Competitive Analysis
Segmentations Covered in Market Analysis:
Customer:
- AMC Clinical
- Community Hospital
- Large Biopharma
- Small Biopharma
- CRO / Reference Labs
Products / Services:
- Scanners / Image acquisition
- Image Management Software
- Image Analysis / AI Algorithms
- Custom Image Analysis Development Services
- Other Services
Histological Method:
- IHC
- H&E
- Immunofluorescence
- In-situ Hybridization
Application:
- Basic & Translational Research
- Clinical Research & Trials
- Routine Clinical
- Training & Slide Archiving
Therapeutic Area:
- Oncology
- Metabolic Disease
- Infectious Disease
- Neurology
- Other
Geography:
- United States
- Europe
- APAC
- Rest of World
Clinical Respondent Survey (N=301)
The focus of this survey was to characterize the use of digital pathology within the clinical lab setting. Though respondents were asked specifically about use of DP within clinical practice, some vendor familiarity may be skewed by research activities. Data presented is enriched for DP users and as a result, survey data tends to skew higher than market averages.
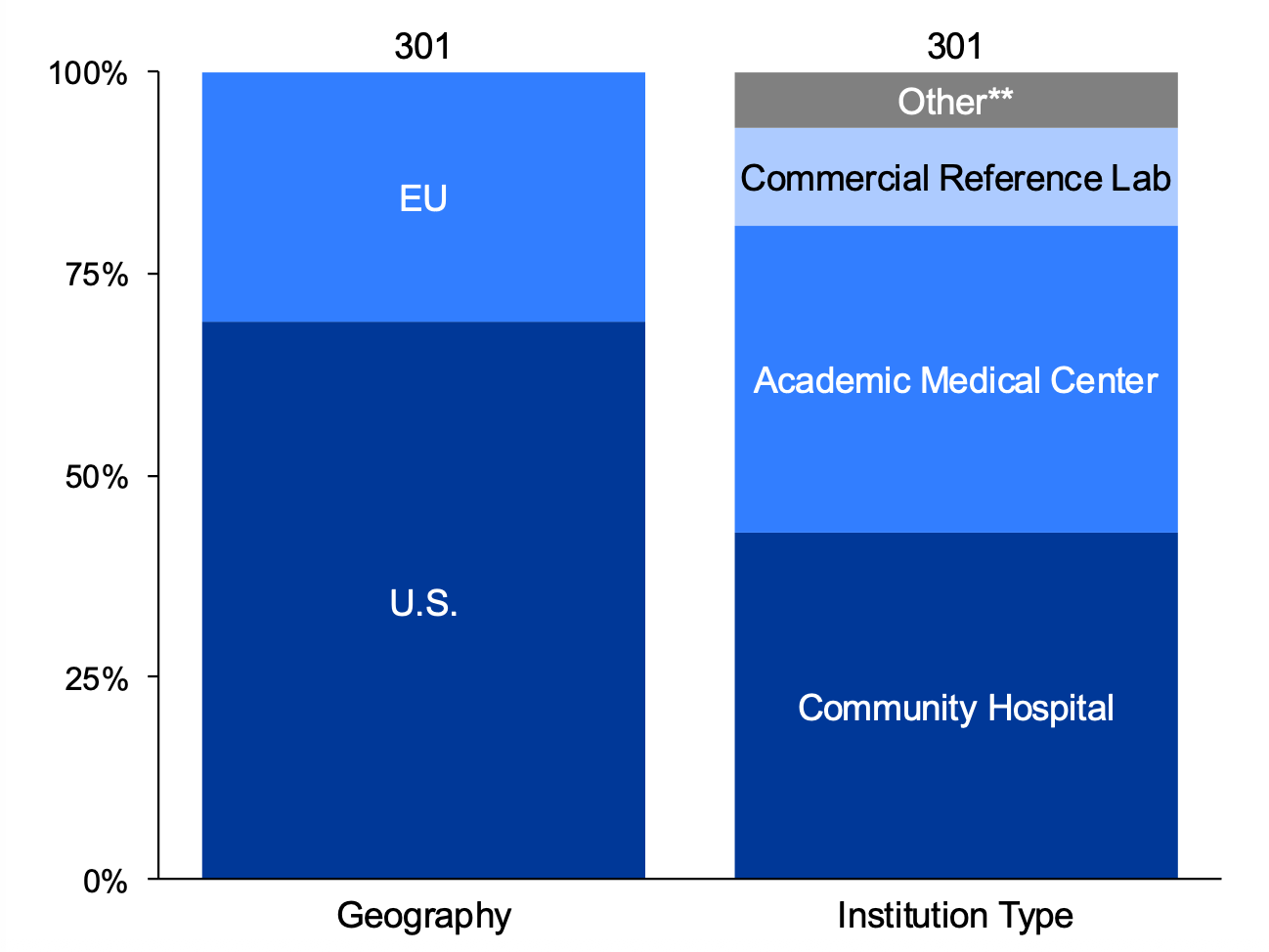
Survey participants were asked about:
- Slide scanners (including use of different vendors, the number of scanners in their lab, utility of scanners, reasons for slide scanning)
- Image management software (including use of different vendors, use cases of IMS)
- IA/ AI algorithms (including use of different vendors, utility of IA / AI tools, reasons for using IA / AI tools, pain points associated with algorithms)
- New technology adoption trends
Purchase Options




Experience the DeciBio Difference
Experience the DeciBio Difference
- $ 8,000.00 USD
- $ 11,500.00 USD
- $ 13,500.00 USD








